Car-T Platform
Advanced immunotherapy that harnesses genetically modified T cells to precisely target and eliminate cancer cells.
Personalized gene cell therapy for the treatment of cancer
CAR-T therapy is a type of immunotherapy where patient's own immune cells are genetically engineered to specifically target cancer cells.


A Breakthrough in Cancer Treatment
CAR-T therapy is a type of cell therapy where genetically modified immune cells from the patient selectively attack cancer cells. 'Kymriah', was approved by the U.S. FDA in 2017 and was the first CAR-T treatment available for patients. From Phase II clinical trials, it boasted nearly 90% complete remission rate and has been called a miracle anticancer drug.
zCAR-T allows for continuous modulation of CAR T-cell activity within patients
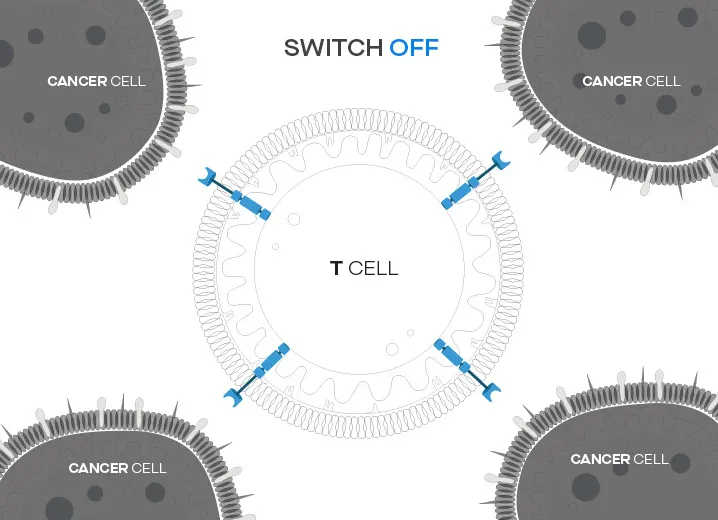
TCT's proprietary zCAR-T treatment is the next generation of CAR-T treatment whose modularity can allow it to be used against various cancer targets as well modulating the activity of CAR-T cells within the patient.
Precision Engineering for Enhanced Safety
Immune cells genetically engineered with zCAR-T are regulated via a switch molecule which modulates not only the activity of the T-cells but also the cancer target. This modularity allows for streamlining production of the treatment as well as minimizing any potential side effects.
Advantages of zCAR-T platform
ON/OFF function
Precise control over T-cell activation for optimal treatment management
Controllable immune response
Adjustable treatment intensity based on patient needs
Expansion of cancer targets
Versatile platform adaptable to various types of cancer
Expansion of treatment possibilities to solid cancer in addition to blood cancer
zCAR-T platform's modular nature means that providing new targeting instructions for zCAR-T cells is as simple as swapping out the switch.
Pipeline derived from CAR-T platform
CAR-T treatment for CD19-positive hematologic cancer cells, developed with in-house NEST platform.
Learn more about AT101